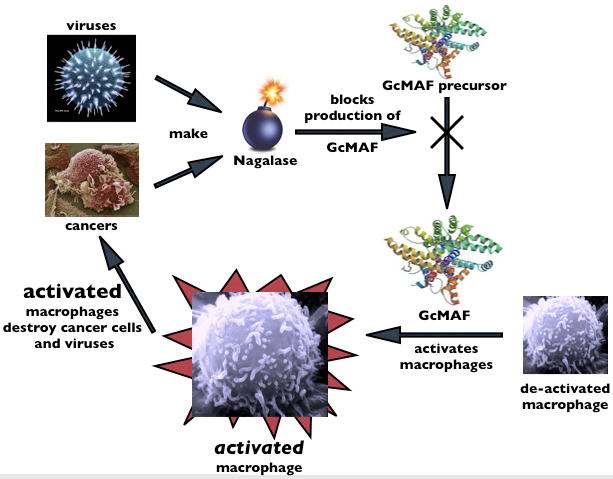
Cancer cells are clever because they disable our immune system
in order to enhance their own survival. Dr. Yamamoto discovered that cancer
cells do this by secreting an enzyme called Nagalase, which prevents the
precursor protein Gc from being converted to MAF. This Nagalase-enzyme
activity can actually be measured in cancer patients; a high nagalase
enzyme reading on a test signifies that you have the very earliest
beginnings of either a cancer or a virus..
Elimination the tumor results in a much lower nagalase reading.
Cancer cells are clever because they disable our immune system
in order to enhance their own survival. Dr. Yamamoto discovered that cancer
cells do this by secreting an enzyme called Nagalase, which prevents the
precursor protein Gc from being converted to MAF. This Nagalase-enzyme
activity can actually be measured in cancer patients; a high nagalase
enzyme reading on a test signifies that you have the very earliest
beginnings of either a cancer or a virus..
Elimination the tumor results in a much lower nagalase reading.
Video below shows how GcMAF eradicates cancer; cells seen via microscopes and time lapse photography in action.
Cancer cells destroyed by First Immune GcMAF (gcmaf.eu)
Source:Immune Biotech Lab: side effects of GcMAF
Explanation about video:
The video illustrates Human MCF7 breast cancer cells both in a corrugated layer on the surface below, and as irregular "fingers" above.
Macrophages activated by First Immune GcMAF are small round circles added at the bottom left.
Time lapse photography over 60 hours shows the cancer monolayer below first changing from corrugated to smooth from the bottom left as the cancer is destroyed; then the cancer "fingers" are also eaten and destroyed by the macrophages.
This video confirms the research paper "Effects of vitamin D-binding protein-derived macrophage-activating factor on human breast cancer cells," published in the journal Anticancer Research 2012 Jan;32(1):45-52.
GcMAF response times: Immune Biotech Lab: GcMAF response times
"GcMAF appears to be successful at rebuilding the immune system in around three weeks in the vast majority - probably over 80%. Remember that since the half life of GcMAF is only one week - you have to keep taking it until your disease has gone, or the immune system gets shut down again. In responders (about 80%) nagalase comes down at the rate of 10-40% every 8 weeks. You should give your immune system 8 weeks for chronic herpes/acne. Allow 24 plus weeks of GcMAF for: Autism (85% improve, 15% eradication), CFS (40% eradication), HIV, Lyme (15% respond, most appear to have the VDR gene blocked and the viruses conceal themselves with biofilms) and stage 1 to 2 cancer, (80% respond). Late stage cancer, perhaps 20% responders, takes a year to 18 months. Remember everyone responds differently. We can't say how you will respond. The more minor the disease, the easier it is for GcMAF and your immune system to eradicate. GcMAF needs normal levels of vitamin D to function strongly. But even in low responders, GcMAF usually appears to stop the advance of cancer."
Since GcMAF is a protein that is dependent on sufficient vitamin D-3 being present in the body, one could infer that those deficient in vitamin D-3 would be prone to cancer and autoimmune disorders! HIV participants who also take 4000IU plus of vitamin D daily can expect to see nagalese drop to normal after 16 weeks.
Research: Research into GcMAF has been occurring since 1990 and hundreds of research papers have explored the role of extracted and purified GcMAF for a variety of conditions. The leading researcher in all of this has been Nobuto Yamamoto. Yamamoto: GcMAF & breast cancer 2008 Yamamoto and other researchers and practitioners have demonstrated that GcMAF can reverse diseases that attack the immune system such as: chronic inflammation, bacterial and viral infections, chronic herpes, chronic acne, Lyme disease, fibromyalgia, osteoporosis, Hodgkin’s, Lupus, MS, Parkinson’s and remarkably – autism. Article by Smith: Grading cancer 2010 is no longer active. Fannon: GcMAF kills prostate cancer Noakes: GcMAF kills cancer cells 2012 Yamamoto: GcMAF & prostate cancer 2008 Yamamoto: lupus & GcMAF 1997 Published patient trials
GcMAF issues related to cancer and other autoimmune disorders have also garnered validity from the attendances of numerous global doctors and researchers attending GcMAP conferences. Although GcMAP had been ignored [ and suppressed ] in the United States since 1990, it is no longer being ignored today by drug companies, the public and medical doctors. Immune Biotech: Conference reports 2013
Because GcMAF is a new approach to fighting cancer and challenges the established ways of fighting cancer, it has been labeled as controversial for the past 20 years. Arney: Controversy GcMAP 2008 McCarty: Early cancer control
Warning: Consult an informed medical healer before using GcMAF, getting tests for GcMAF or Nagalase. Many medical doctors are NOT informed about GcMAF so you need to find a medical doctor who is informed and trained in the use of GcMAF! A search on the internet reveals that GcMAF is already being marketed and may be purchased by individuals. Immune Biotech Lab: side effects of GcMAF Likewise, you can obtain blood tests for GcMAF and nagalase. Testing GcMAF is a time consuming, highly specialized and expensive process. According to Immune Active Lab, "the only way to test that GcMAF is actively with live macrophage and live cancer cell lines. In the laboratory tests we do on our GcMAF batches, we photograph through microscopes as newly activated macrophages eat cancer cells, and in the absence of macrophages, our GcMAF turns the cancer cells back into healthy cells. That is how we know a batch is active." Other companies marketing GcMAF have not disclosed their product verification process. GcMAF needs to be purified every time prior to using it. The real full, safe, universal, and clinical application of this new medical-health marvel may still be a few years away. This commentary about verifiying an active working synthesis of GcMAF is not an endorsement of Immune Biotech Lab products.
Possible GcMAF complications can happen and why you need to be under the care of an informed medical doctor: IRIS is a disease that attacks the immune system the moment it tries to repair itself. IRIS is a poorly understood, acute illness called Immune Reconstitution Inflammatory Syndrome (IRIS) that develops in some HIV-infected individuals soon after they begin anti-retroviral therapy. IRIS affects certain HIV-infected individuals whose immune systems are heavily damaged by the virus and who have a treated or undiagnosed AIDS-associated infection. When these individuals start anti-retroviral therapy and their immune cells begin to regenerate, the immune system unexpectedly produces an exaggerated response that unmasks or worsens the symptoms of the co-infection.
Testing of Nagalase to Determine if GcMAF Should Be Considered: Since viruses and diseased cells release negalase, people who have a higher level of negalase in their body means that the immune system is not effective at dealing with invaders. Children with autism typically have nagalase levels triple the normal level. Nagalase blood tests are available so that the blood level number can be monitored to see the impact that GcMAF is having on the immune system. Typically if the number of nagalase in the blood is elevated, this means that GcMAF is needed to assist the immune system to get back into balance. People with normal nagalase levels typically will not benefit from GcMAF, so it is wise to get a blood test before treatment as well as periodically while on GcMAF. According to research, nagalase levels can be cut in half every eight weeks while on GcMAF." Article by Smith: GcMAF Book Table of contents is no longer active.
Testing for Cancer:
The only test on the market now to find tumor cells in blood – CellSearch, made by J&J's Veridex unit – just gives a cell count. It doesn't capture whole cells that doctors can analyze to choose treatments. Article by Marchione: New blood test is no longer active. Interest in trying to collect these cells soared in 2007, after Haber and his colleagues published a study of Mass General's test. Noakes: GcMAF kills cancer cells 2012 It is far more powerful than CellSearch and traps cells intact. It requires only a couple of teaspoons of blood and can be done repeatedly to monitor treatment or determine why a drug has stopped working and what to try next. The new test is (a) in need of a lot of further study and (b) very expensive.
AMAS test is similar in a way. AMAS is much cheaper and — best of all — available now. Unlike tests such as CEA [Carcinoembryonic antigen is a glycoprotein involved in cell adhesion. In adults, an abnormal amount of CEA may be a sign of cancer.] which measure less well-defined antigens whose serum levels tend to be inconstant but elevated late in the disease. The AMAS test measures a well-defined antibody whose serum levels rise early in the course of the disease.
The AMAS test is controversial. AMAS stands for anti-malignin antibody in serum. The AMAS test looks for the presence of a substance called "malignin" in the body by detecting antibodies to it in the blood. Supposedly, malignin is a marker for any type of cancer. "The AMAS test is useful both as a screening test for early cancer and for monitoring cancer therapies. AMAS is elevated when cancer is present and goes down below baseline when cancer is gone. AMAS is over 99% accurate [ when done twice ] and can be used instead of Nagalase to find and follow cancers. [ Unlike Nagalase, AMAS does not detect the presence of viruses.] AMAS is both a cancer screening test and a cancer monitoring test. .... AMAS differs from Nagalase in that AMAS is specific for cancer, while Nagalase identifies an enzyme made by both viruses and cancer cells and is therefore unable (by itself) to distinguish between the two. Because Nagalase testing cannot distinguish the difference between cancer and virus, AMAS is a better screening test for cancer." Article by Smith: GcMAF Book Table of contents is no longer active.
The Future: GcMAF is a naturally made molecule and is not patentable, though its manufacturing process is patent protected. There is no evidence of any current effort to commercialize this therapy or put it into practice.
The National Cancer Institute is totally silent on this discovery and there is no evidence the cancer care industry plans to quickly mobilize to use this otherwise harmless but effective treatment. The truth: the silence from the National Cancer Institute and National Institutes of Health on this potential therapy implies that both are suppressing GcMAF clinical research and application.
What can you do?
Demand that your congressperson and senator take immediate action:
- Pass immediate legislation to provide adequate funding to validate clinical trials and more research for GcMAF.
- Request that President Obama direct telemedia to promote this proposed research for auto-immune disorders and cancer.
- Demand that there be continuous follow-up on the progress of PcMAF.
- Start a petition [ public demand ] to gather signatures to force politicians and the White House to take immediate action.
References:
Arney Kat, “Cancer cured for good?” – Gc-MAF and the miracle cure," Cancer Research UK, December 3, 2008. Arney: Controversy GcMAP 2008
Cost of GcMAF therapy: Nothing currently prevents cancer patients, on their own initiative and at their own expense, from acquiring GcMAF for personal use. Currently, GcMAF acquired from a European source costs about $600 per month to use if 100 ng GcMAF is injected weekly.
Doctors working with GcMAF in a clinical setting in Europe: [incomplete ]
Prof Dr Thomas Vogel, University of Frankfurt Cancer Clinic. (reputedly one of Germany’s well known cancer specialists) Theodor-Stern Kai 7,
60590 Frankfurt tel.: 069-6301-4736 Fax: 069- 6301-7288Dr Uta Santos Koenig 0043 6642829909 Albertplatz 8a, 1080 Wien Vienna, Austria
Dr Martin von Rosen Zentrum für Bilogische Krebsmedizin, Schlossparkklinik Schloßplatz 3, 36129 Gersfeld Rhon, Germany tel.: +49 (0)6654-960950
Dr Finn Scott Andersen Humlegaarden Humlebæk Strandvej 11 DK – 3050 Humlebæk Tel.:(+45) 49 13 24 65
Dr. Henning Saupe Arcadia-Praxisklinik, Germaniastrasse 1a D-34119 Kassel Germany Tel:+49 561 3168901 Fax: +49 561 3168902
Hufeland klinik. Gabriele Wöppel, Angelika Wöppel Löffelstelzer Str. 1-3 D-97980 Bad Mergentheim Tel:+49-7931-536-0 Fax: +49 -7931-536-333
Dr Migeod, sekretariat, Biomed BioMed Fachkliniak fr Onkologie Immunologie und HyperthermieTischberger Strae 5+8 D-76887 Bad Bergzabern Telefon +49 (0) 6343 -705 – 0 Telefax +49 (0) 6343 – 705 – 913
Dr Ashhoff, Villa Medic. Dr. Burkhard Aschhoff Privatklinik Villa Medica Klosterstrasse 205,?67480 Edenkoben tel.: +49 6323 8020 Fax: +49 6323 7943
Fannon Michael and others, "Vitamin D Binding Protein-Macrophage Activating Factor Directly Inhibits Proliferation, Migration, and uPAR Expression of Prostate Cancer Cells," PLOS ONE, October 18, 2010. [ PLOS ONE (eISSN-1932-6203) is an international, peer-reviewed, open-access, online publication. Co-authored paper which demonstrated the ability of GcMAF to directly kill prostate cancer cells in vitro.] Fannon: GcMAF kills prostate cancer
GcMAF is glycoprotein macrophage activating factor.
GcMAF Research, Published patient trials, FirstImmune GcMAF. Published patient trials
GcMAF research papers, February 2013. GcMAF research papers List on the US Library of Medicine, 142 eminent scientists from 8 nations have published 59 major GcMAF research papers, which can be viewed on the US Government's Pubmed system.
Gregory KJ, Zhao B, Bielenberg DR, Dridi S, Wu J, et al. (2010) Vitamin D Binding Protein-Macrophage Activating Factor Directly Inhibits Proliferation, Migration, and uPAR Expression of Prostate Cancer Cells. PLoS ONE 5(10): e13428. doi:10.1371/journal.pone.0013428, October 18, 2010. Gregory: 2010
Immune Biotech Labs, "GcMAF - for cancer, infections, viral and immune diseases," First Immune GcMAF, 2013. Immune Biotech Lab: GcMAF response times
"The many scientists who have published papers on trials of GcMAF selected those in the early stages of cancer and HIV, and reported nearly 100 percent success, with no recurrence after many years. They did not attempt trials on people with large tumours.
Our trials are quite different: many people are over 50, some over 80, with advanced or terminal cancers, with significant tumour mass.
Our GcMAF appears to be successful at rebuilding the immune system in around three weeks in the vast majority - probably over 80%. Remember the half life of GcMAF is only one week - you have to keep taking it until your disease has gone, or the immune system gets shut down again. In responders (about 80%) nagalase comes down at the rate of 10-40% every 8 weeks.
You should give your immune system 8 weeks for chronic herpes/acne. Allow 24 weeks plus of GcMAF for: Autism (85% improve, 15% eradication), CFS (40% eradication), HIV, Lyme (15% respond, most appear to have the VDR gene blocked and the viruses conceal themselves with biofilms) and stage 1 to 2 cancer, (80% respond). Late stage cancer, perhaps 20% responders, takes a year to 18 months. Remember everyone responds differently. We can't say how you will respond.
The more minor the disease, the easier it is for GcMAF and your immune system to eradicate. GcMAF needs normal levels of vitamin D to function strongly. But even in low responders, GcMAF usually appears to stop the advance of cancer."
Immune Biotech Ltd.,"Immuno Biotech presents GcMAF Research Papers on fighting Cancer and Autism treatment at International Conferences in the USA and Dubai," RealTime press release, February 08. Immune Biotech: Conference reports 2013
Immune Biotech Ltd., "Side Effects," FirstImmune [lab]. Immune Biotech Lab: side effects of GcMAF Location: Clos de Balade 21, 1140 Evere, Brussels, Belgium. Phone: 44 7781 411 737.
Toshio Inui, Daisuke Kuchiike, Kentaro Kubo, Martin Mette, Yoshihiro Uto, Hitoshi Hori, and Norihiro Sakamoto , "Clinical Experience of Integrative Cancer Immunotherapy with GcMAF," ANTICANCER RESEARCH, June 3, 2013, 33: 2917-2920 [Japan]. TOSHIO: Clinical experience 2013
Macrophages (meaning “big eaters” in Greek) are an important type of white blood cell. They patrol the body, eating up foreign invaders and dead cells. They also help to alert other immune cells to the presence of infections.
Marchione Marilynn, "A New Blood Test To Detect Cancer Could Be Coming To Your Doctor's Office," The Huffington Post, January 03, 2011. Article by Marchione: New blood test is no longer active.
"The only test on the market now to find tumor cells in blood – CellSearch, made by J&J's Veridex unit – just gives a cell count. It doesn't capture whole cells that doctors can analyze to choose treatments.
Interest in trying to collect these cells soared in 2007, after Haber and his colleagues published a study of Mass General's test. It is far more powerful than CellSearch and traps cells intact. It requires only a couple of teaspoons of blood and can be done repeatedly to monitor treatment or determine why a drug has stopped working and what to try next."
McCarty Mark F., "Overview of Macrophage Activating Factor and the Nagalase Assay – Potential for Control of Micrometastatic or Early Primary Cancer," Catalytic Longevity. McCarty: Early cancer control
Noakes David, "Cancer cells destroyed by First Immune GcMAF (gcmaf.eu)," Immune Biotech Ltd., UTube, October 21, 2012. Noakes: GcMAF kills cancer cells 2012
Smith, Timothy J. M.D., "Chapter 1 A Cure for Metastatic Cancer?" The GcMAF Book, Copyright © 2010. Article by Smith: GcMAF Book Table of contents is no longer active. Article by Smith: Cancer cure is no longer active.
Smith, Timothy J. M.D., "Chapter 17 Differentiation and Cancer Grading," The GcMAF Book, Copyright © 2010. Article by Smith: Grading cancer is no longer active.
Yamamoto Nobuto, Hirofumi Suyama, and Nobuyuki Yamamoto, "Immunotherapy for Prostate Cancer with Gc Protein-Derived Macrophage-Activating Factor, GcMAF1," Transl Oncol. July 2008; 1(2): 65–72. Yamamoto: GcMAF & prostate cancer 2008 Procedure for preparing GcMAF; GcMAF Therapy for Prostate Cancer Patients; This investigation was supported in part by the US Public Health Service Grant AI-32140 and by an Elsa U. Pardee Foundation grant.
Yamamoto N, Suyama, H, Yamamoto, N, Ushijima N., "Immunotherapy of metastatic breast cancer patients with vitamin D-binding protein-derived macrophage activating factor (GcMAF)," Int J Cancer, January 15, 2008;122(2):461-7. Yamamoto: GcMAF & breast cancer 2008
Yamamoto N. et al. , "Deglycosylation of serum vitamin D3-binding protein by alpha-Nacetylgalactosaminidase detected in the plasma of patients with systemic lupus erythematosus," Clin. Immunol Immunopathol, March 1997; 82(3):290-8. Yamamoto: lupus & GcMAF 1997
"Gc protein is the precursor for MAF. Treatment of Gc protein with immobilized β-galactosidase and sialidase generates a remarkably high titered macrophage-activating factor (GcMAF). When peripheral blood monocytes/macrophages (designated macrophages) of 33 systemic lupus erythematosus patients were incubated with GcMAF (100 pg/ml), the macrophages of all patients were activated as determined by superoxide generation. However, the precursor activity of patient plasma Gc protein was lost or reduced in these patients. .... Thus, elevated plasma α-N-acetylgalactosaminidase activity resulting in the loss of MAF precursor activity and reduced macrophage activity may play a role in the pathogenesis of systemic lupus erythematosus."
Yamamoto N, Urade M., "Pathogenic significance of alpha-N-acetylgalactosaminidase activity found in the hemagglutinin of influenza virus," Microbes Infect. April 7, 2005, (4):674-81.
Wikipedia,"GcMAF." Wiki: GcMAF