Compiled by Walter Sorochan Emeritus Professor San Diego State University
Posted December 12, 2015; Updated November 19, 2021. Disclaimer; The information presented here is for informative and educational purposes only and is not intended as curative or prescriptive advice. The statements of this web-site have not been evaluated by the United States Food and Drug Administration (FDA). Nothing stated here should be considered as medical advice for dealing with a given problem, or to diagnose / treat / prevent / cure any disease. This is work in progress.
|
Perspective: This article attempts to put the media hype about keytruda into perspective. Keytruda is a drug that may have a lot of uncertainty as well as controversy. The author's attempt is to present the information in a neutral and realistic analysis. Search for the truth! The author has no vested interests, other than having Actinic Keratosis skin cancer. |
The exciting health news, during weekend of December 4 to 6, 2015, was that there was supposedly a new drug that destroyed former President Jimmy Carter's cancer. Not exactly a sure cure but giving us hope that cancer can finally be controlled. But was this more media hype than truth? Let's take a closer look at this news.
"Jimmy Carter credits the new cancer drug Keytruda for shrinking his brain tumors completely. It's one more possible victory for the newest class of cancer drugs that empower the immune system to fight off tumors. But cancer experts note that it's not at all clear that the drug itself is what shrank Carter's tumors. The former president was also treated with radiation to his brain and had a large tumor on his liver removed. While most immune therapy drugs boost the immune system so that it can battle the cancer, Keytruda was the first drug to take a different approach that disrupts a trick tumors use to hide from immune cells." Fox: Carter tumor vanish 2015
"Keytruda — known generically as pembrolizumab — targets the activity of genes called PD-1 and PD-L1. The interaction between the two genes lets some tumors escape detection and destruction by immune system cells. PD-1 stops immune cells from attacking normal healthy cells by mistake. Tumor cells make PD-L1 turn on PD-1 when immune cells approach. Keytruda, an engineered immune protein called a monoclonal antibody, disrupts this cloaking effect and lets the immune cells do their job and eat the tumor cell." Fox: Carter tumor vanish 2015
Below is a short video explaining how keytruda activates the immune system to fight cancer [ By Cancer Research Institute ] 1:51 mns:
A Quick Realistic Outlook: The theory behind immunotherapy has been around for decades, but it is only in very recent years that scientists have been able to put it into practice with, so far, just a couple of remarkably successful drugs.
So what are we to understand from this medical media report? Medical doctors admit that is not clear that keytruda was responsible for shrinking or getting rid of President Carter's cancer. Indeed, common sense should remind you that there were a lot of other possible factors not mentioned in the report that may have been helpers in destroying Carter's cancer. Did the food Carter ate have an impact on his cancer? And how about the placebo effect? And let us not forget that the survival rate among those using keytruda therapy is very low at this time. There is also speculation that keytruda may not cure cancer and that cancer may reappear after initial disappearance of cancer. A cancer patient may need to take keytruda for the rest of her/his life for the current price of $ 10,000 every month. Scientists just do not have all the data about keytruda at this time.
Medical science does not have all the answers as to what caused Carter's cancer to go into remission, although the drug media wants to take credit for Carter's cancer supposedly going into remission. Doctors admit that it is not clear what caused the disappearance of cancer. But the drug company, Merck, who developed keytruda has claimed credit for the cancer disappearing. Merck charges $10,000 for less than a month treatment; a cost that is out of reach for most cancer patients. Not exactly a humane approach. This is typical of "tooting your own horn" and taking all the credit and money!
What is germaine in the media report is the recognition idea that it is the immune system that can destroy cancer and heals the body. Such recognition goes back thousands of years to the Greek physician, Hypocrates, who recognized that: "Food is your best medicine!" Unfortunately, most medical doctors have not had nutrition in their medical training, so it should not be surprising that all of a sudden they are becoming aware that food may be a silent helper in healing the body.
Today, new research about the biochemistry of the body and how it works puts the medication keytruda into perspective. Methylation and methyl groups found in foods help bolster the immune system that, in turn, originates in the large colon that, in turn, is controlled by bacteria in the colon. It is the kind of food we eat that controls the good and bad bacteria in the colon. Good bacteria supercharges the immune system while bad bacteria cause disease. The addage 'you are what you eat' is true!
In spite of the many shortcomings of keytruda, if it shrinks the cancer tumor, it appears to be working and this in itself is a good sign. As a cancer patient, you may not be cured but the tumor is in some degree of remission. You would have a large part of your life restored, you regain hope and all this is great. The video below gives more information about how a patient at UCLA hospital who was helped [ 3.52 mns.]:
Readers who may have cancer need to read the information below to become informed and decide what is best for them. Ask your therapist what your options are and get a second and third opinion.
Below is more information about the details of krytruda that is good to know and may help you decide about using it:
History of cancer fighting drugs:
"The drug keytruda, is part of a new class of drugs that fight cancer by boosting the body's immune system. Keytruda, which costs $150,000 a year, is one of eight new melanoma drugs approved since 2011," said Timothy Turnham, executive director of the Melanoma Research Foundation. Although none of the new drugs has been proven to cure cancer, their arrival was welcome news to patients and cancer advocates. zabo Carter cancer in remission :2015
"The first cancer drugs, developed in the 1940s, were chemotherapies, which kill growing cells, Shepard said. While these therapies can kill cancers — which grow rapidly — they also kill normal growing cells, such as the cells lining the gut or blood cells. That's why people treated with chemotherapy often develop anemia, nausea and vomiting, and can be vulnerable to life-threatening infections.
In 1998, the Food and Drug Administration approved the drug Herceptin for breast cancer. It was part of a new breed of "targeted therapies," designed to kill cancer cells, rather than all growing cells. That was followed in 2001 by the approval of Gleevec — one of the few real wonder drugs in cancer — for chronic myeloid leukemia. Scientists were able to design these cancer-specific drugs because of a greater understanding of the genes that drive cancer.
But targeted therapies have their own limitations, Shepard said. Although targeted therapies can block parts of cancer cells that promote growth, tumor cells often adapt, finding a "workaround" that allows them to begin growing again.
The new immune therapies don't kill cancer cells directly. Instead, they aim to rev up the immune system, Shepard said. Although the immune system can reliably recognize and kill invaders like bacteria and viruses, the immune system often looks the other way on cancer cells, which don't appear foreign. That's because cancers don't come from outside the body; they are malignant versions of a patient's own cells.
Immune therapies aim to "unmask" cancer cells, so that the body recognizes them as the dangerous killers that they are, Shepard said." Szabo Carter cancer in remission :2015
 The Food and Drug Administration gave Keytruda accelerated approval in 2014 for patients, like Carter, whose melanoma has spread. It was on the basis of a big study that showed Keytruda could shrink the tumors of about a third of patients by as much as 90 percent.
Fox: Carter tumor vanish 2015
The Food and Drug Administration gave Keytruda accelerated approval in 2014 for patients, like Carter, whose melanoma has spread. It was on the basis of a big study that showed Keytruda could shrink the tumors of about a third of patients by as much as 90 percent.
Fox: Carter tumor vanish 2015
How the drug works? Keytruda works by way of immunotherapy. Keytruda® is a highly selective humanized monoclonal IgG4 antibody directed against the PD-1 receptor on the cell surface. The antibody [drug] blocks the PD-1 receptor, preventing binding and activation of PD-L1 and PD-L2. This mechanism causes the activation of T-cell mediated immune responses against tumor cells.
"In clinical trials, anti–PD-1 and anti–PD-L1 antibodies produce durable responses in approximately 20% of unselected patients with advanced non–small-cell lung cancer." Garon: Pembrolizumab safety treating small cancer 2015
"Keytruda is the brand name for a generic drug known as pembrolizumab. That drug targets the activity of two specific genes: PD-1 and PD-L1. Interactions between these two genes allow some tumors to escape destruction and detection by immune system cells, which then lets cancer spread throughout the body. Basically, PD-1 prevents immune system cells from attacking normal healthy cells accidentally. Cancer cells make PD-L1 turn on PD-1 when immune cells approach. Keytruda reverses this mechanism by acting as a monoclonal antibody, which is a type of engineered-immune protein. This disrupts the “cloaking” effect used by tumor cells, which then lets your immune system do its job and fight back against the tumor cells."
"Keytruda works by targeting the cellular pathway known as PD-1/PD-L1 (proteins found on the body’s immune cells and some cancer cells). By blocking this pathway, Keytruda may help the body’s immune system fight the cancer cells. In 2014, Keytruda was approved to treat patients with advanced melanoma following treatment with ipilimumab, a type of immunotherapy. Another drug, Opdivo (nivolumab), manufactured by Bristol-Meyers Squibb, also targets the PD-1/PD-L1 pathway and was approved to treat squamous non-small cell lung cancer (a certain kind of NSCLC) in 2015." Article by FDA: Approves Keytruda 2015 is no longer active.
Below is an easy to understand video about the process of immunotherapy [Society for autoimmunotherapy of cancer, 11:52 mns]:
Keytruda side effects may include: Drugs: Keytruda Article by FDA: Approves Keytruda 2015 is no longer active.
- nausea
- loss of appetite
- diarrhea
- constipation
- itching or rash
- cough
- joint pain
- feeling tired
This is not a complete list of side effects and others may occur.
Survival rate: There are testimonials that some cancer patients receiving keytruda do survive for longer than two years. But this information is not verified. Survival rate for cancer patients being treated with keytruda at this time is virtually unknown since this treatment is so new. Doctors are not even sure that those being treated are surviving because of keytruda, or of other medications or factors. Survival among patients with advanced melanoma who were given pembrolizumab were good at 12 months – 74% given doses every two weeks were still alive a year later. Szabo Carter cancer in remission :2015
Source: Garon: Pembrolizumab safety treating small cancer 2015
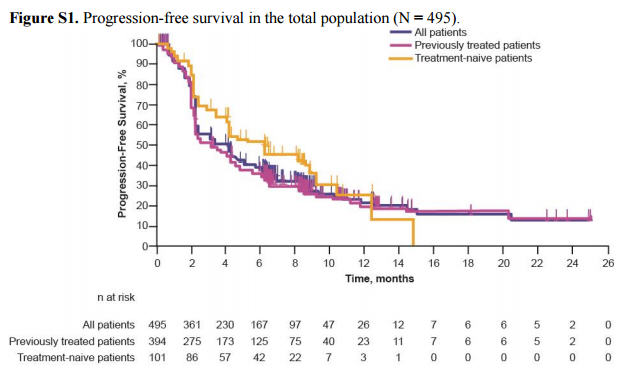
It was on the basis of a big study that showed Keytruda could shrink the tumors of about a third of patients by as much as 90 percent. Fox: Keytruda another look 2015
In spite of the lack of good long-term survival and cure rates, cancer patients should have an open mind to using keytruda or similar medications. It may be only a matter of time before the researchers get it right! Do not lose hope!
KEYTRUDA USE: is a prescription medicine used to treat a kind of skin cancer called melanoma: Merek: Medication Guide 2015
- has spread or cannot be removed by surgery (advanced melanoma)
- after you have tried a medicine call ipilimumab and it did not work or is no longer working
- if your tumor has an abnormal “BRAF” gene, and you also have tried a different medicine called a BRAF inhibitor, and it did not work or is no longer working.
KEYTRUDA may be used in a kind of lung cancer called non-small cell lung cancer: Merek: Medication Guide 2015
- has spread
- tests positive for “PD-L1”
- you have tried chemotherapy that contains platinum, and it did not work or is no longer working
- if your tumor has an abnormal “EGFR” or “ALK” gene, and you have also tried an EGFR or ALK inhibitor medicine.
It is not known if KEYTRUDA is safe and effective in children less than 18 years of age.
Biomicrobia, keytruda and cancer research:
Medical researchers and the general public may be skeptical about feces being linked to health and disease, but the research in the past 15 years says otherwise. Below are just two such researches:
"Sivan and colleagues compared genetically identical mice obtained from two different mouse facilities that had different gut microbiota. Interestingly, these mice reacted differently to syngeneic implanted mouse melanoma tumors, apparently because of differences in the vigor of their anti-tumor T-cell responses. These differences were eliminated by cohousing and could be reversed by fecal transplantation from the mice with anti-tumor immunity, implicating the gut microbiome. Furthermore, fecal transfer augmented responses to anti–PD-L1 immunotherapy. Following sequencing of the bacteria, they found that Bifidobacterium species were overrepresented in mice with better anti-tumor immunity, and introduction of those species into the other mice produced anti-tumor responses." Sivan: Bacteria promote anti cancer 2015 Tsai: 2015
"Vétizou and colleagues linked anti-CTLA4 therapy to human responses to melanoma therapy. Anti-CTLA4 therapy can produce adverse gastrointestinal (GI) tract reactions with the development of antibodies to gut flora. They found that anti-CTLA4 therapy decreased numbers of Bacteroidales and Burkholderiales species and increased Clostridiales species in feces. Patients treated with ipilimumab for melanoma also showed decreased Bacteroidales species in their stool. When this stool was transplanted into mice, it enhanced responses to anti-CTLA4 therapy, and direct inoculation of the relevant Bacteroidales species increased melanoma-specific memory T-cell responses." Vétizou: Cancer immunity needs gut microbiota 2015 Tsai: 2015
Two groundbreaking studies implicate specific bacterial species in regulating gut immunity and response to immunotherapy in physically distant tumors — an entirely new approach in cancer therapy. The influence of the microbiome on cancer susceptibility and therapy effectiveness has been shown. Furthermore, commensal organisms have critical roles in tuning immunity at epithelial surfaces, suggesting their potential role in regulating immunotherapy response. Tsai: 2015
COMMENT: These technically difficult and groundbreaking studies implicate specific bacterial species in regulating gut immunity and responses to immunotherapies in physically distant tumors. This approach presents an entirely new approach to cancer therapy. How important the gut microbiome is as a determinant of response to therapy is not yet known, but immunotherapies may be optimized by manipulation of the GI microbiome in the future. Tsai: 2015
|
Importance of methylation to cancer "Cancer researchers are just beginning to understand the ramifications of the fundamental discoveries behind the 2015 Nobel Prizes in Chemistry, which were awarded to three scientists for explaining how cells repair mistakes in DNA that occur when cells divide. When deoxyribonucleic acid (DNA) repair mechanisms fail, they predispose people to cancer. That is especially true of individuals with mismatch repair defects, a mechanism discovered by Paul Modrich of Duke University and the Howard Hughes Medical Institute. Modrich, Tomas Lindahl and Aziz Sancar won the prize for "mechanistic studies of DNA repair." Their work mapped how cells repair DNA to prevent damaging errors from appearing in genetic information. Subsequent work by Dr. Bert Vogelstein of the Johns Hopkins Kimmel Cancer Center and Richard Kolodner, then at Harvard Medical School and currently at the University of California, San Diego, showed mismatch repair defects are the chief cause of the most common inherited form of colorectal cancer, affecting 15 percent of colon cancer patients. These defects may now help predict which patients are most likely to benefit from immunotherapies, promising new drugs that enlist the immune system to fight cancers. A small study published earlier this year in the New England Journal of Medicine showed that 92 percent of patients with advanced colon and rectal cancers who had mismatch repair defects responded to Merck & Co's immunotherapy drug Keytruda. That compared to a response of 16 percent in patients with the same cancers who did not carry the defect. The findings could also be applicable to patients with other cancers who have the same DNA repair defects, and possibly to those with defects in other DNA repair mechanisms as well, Vogelstein said. "Mismatch repair defects are found not only in hereditary colorectal cancers. They are found in about 2 percent of cancer patients overall," he said in a telephone interview. Vogelstein also said mismatch repair is a marker for response to anti-PD-1 inhibitor drugs such as Keytruda because patients with mismatch repair defects have tumors teeming with mutations - far more than cancer patients with functioning DNA repair genes. Since the immune system is trained to recognize foreign invaders, Hopkins researchers believe immune-boosting drugs such as Keytruda or Bristol-Myers Squibb's Opdivo, will perform better in mutation-loaded tumors. So far, the findings, while strong, are preliminary. Vogelstein said if this same strategy extends to other cancer types, "it could provide a very useful therapy for as many as one in 50 patients with cancer worldwide." Dr. Roger Perlmutter, president of Merck Research Laboratories, said "every company that's interested in making drugs that enhance the ability of the immune system to recognize cancer is interested" in exploring DNA repair mechanisms. "In colorectal cancer, it's unambiguous. Administration of Keytruda is much, much more effective in those people who have DNA repair mutations," he said. The company is conducting a larger clinical trial in hopes getting FDA approval for the drug in colorectal cancers. Keytruda is already approved in melanoma, and in some forms of advanced lung cancer." Nobel prize & immunotherapies |
Final thoughts: Don't give up hope. Go for it! The video below says it best [ 4:12 mns]:
In light of the current limitation of keytruda .... like how expensive it is for most cancer patients, cancer patients should consider other adjunct options like methylation, ingesting seeds and whole plant foods to help at keep cancer in check.
References
Keytruda – Immunotherapy Drug Guide. Keytruda guide
FDA, "FDA approves Keytruda for advanced non-small cell lung cancer," U.S. Food and Drug Administration, October 2, 2015. Article by FDA: Approves Keytruda 2015 is no longer active.
Drugs.com, "Keytruda," Drugs,com, Drugs: Keytruda
Fox Maggie, "Here's a Look at Keytruda, the Drug Jimmy Carter Says Made His Tumors Vanish," NBC News, December 7, 2015. Fox: Carter tumor vanish 2015
Fox Maggie, "Here's a Look at Keytruda, the Drug Jimmy Carter Says Made His Tumors Vanish," NBC News, December 7, 2015. Fox: Keytruda another look 2015
Garon Edward B., and others, "Pembrolizumab for the Treatment of Non–Small-Cell Lung Cancer," N Engl J Med, May 21, 2015; 372:2018-2028. Garon: Pembrolizumab safety treating small cancer 2015
Sivan A et al. "Commensal Bifidobacterium promotes antitumor immunity and facilitates anti–PD-L1 efficacy," Science, November, 27,2015; 350; 1084. Sivan: Bacteria promote anti cancer 2015
Vétizou M et al., "Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota," Science, November 27,2015 350, 1079. Vétizou: Cancer immunity needs gut microbiota 2015
Merek, "MEDICATION GUIDE KEYTRUDA® (pembrolizumab) injection," Merek Oncology, 2015. Merek: Medication Guide 2015
"Nobel discoveries on DNA repair now fueling cancer drug research," October 08, 2015. Nobel prize & immunotherapies
Szabo Liz, "Jimmy Carter's remission shows power of new cancer drugs," USA TODAY, December 7, 2015. Szabo Carter cancer in remission :2015
Tsai Kenneth Y., reviewing Sivan A et al., "Melanoma Meets Microbiome," NEJM, mber 9, 2015. Tsai: 2015;