By Walter Sorochan: Emeritus Professor San Diego State University
Posted July 24, 2015; updated October 23, 2021. Disclaimer ; The information provided below is intended for educational purposes only. It is not meant to either directly or indirectly give medical advice or prescribe treatment.
Health care is about to change dramatically in the next 10 years. Not only will doctors and hospitals need to change how they take care of their patients, but you will also be able to enjoy more and better health care and prevent many of today's debilitating diseases and illnesses. Indeed, new technology may even make some doctors and hospitals obsolete and extend life beyond 100 years! You will need to take care of your health into your hands, and prevent chronic diseases and disorders by making changes to your lifestyle. This is the future! This article briefly summarizes all these emerging new trends.
Lets start with the early supporting research indicating such trends.
It was the German monk Gregory Mendell whose fascination with peas ignited interest in genetics and that we all inherited characteristics from our ancestors. Though farmers had known for centuries that crossbreeding of animals and plants could favor certain desirable traits, Mendel's pea plant experiments conducted between 1856 and 1863 established many of the rules of heredity, now referred to as the laws of Mendelian inheritance.
Years later, anatomist Leonard Hayflick in 1961, at the Wistar Institute in Philadelphia, Pennsylvania, demonstrated that a population of normal human fetal cells in a cell culture will divide between 40 and 60 times. This discovery .... that a cell dies when the length of it's telomere [ a cap at the end of a cell] is depleted, became known as the "Hayflick Limit.” These observations gave way to thinking that we inherit good and bad genes, how long we would live and that we are stuck with predispositions to diseases and disorders of life. That is, if your ancestors had cancer, then you might also get cancer. But this kind of thinking is about to change.
Many studies since 1960 attempted to verify the Hayflick limit and try to extend the Hayflick limit. Research focused on the impact of nutrition, pollution and life style on wellbeing and longevity, laying the foundation to the more game-changing studies that have followed since 2000.
For example, Colin Campbell, professor of Nutritional Biochemistry at Cornel University and his colleagues conducted a 27-year China Project research that found people who ate a high amount of animal meat were most susceptible to cancer. Protein is needed for body activity but when you eat above the required amount of approximately 10 %, then you turn on cancer tumor growth. Changing one's high meat diet can also dramatically reduce the risk of diabetes, heart disease, auto-immune disorders and obesity. These findings are significant because they triggered more supporting research that confirmed what most astute persons already knew; that a healthy life style can prevent chronic diseases, extend life and lower the cost of health care.
Recent important research has focused on many studies related to bacteria in the the digestive system that are linked to the functioning of the immune system.
Then there is the study of Epigenetics, the powerful way that food, environment, thoughts and lifestyle impact on our genes and cause the genes to change how they work [ but not change the genes themselves ].
Dean Ornish explored lifestyle interventions Ornish: Lifestyle intervention & heart disease 1998 , and Carol Grieder and her colleagues opened the fountain of youth for discovery and work on telomerase, the enzyme that lengthens telomeres and extends life; findings were published in 1985 in the journal, Cell Wade: Nobel prize Telomerase 2009 Activating the enzyme, telomerase, protects the telomeres from shortening and serves as an anti-aging treatment, slowing or reversing aging. On the contrary, knocking out or inhibiting telomerase activity allows telomeres to shorten and accelerate aging.
Telomeres are tiny units [like caps we put on our heads] of DNA at the very end of each chromosome, these shorten with time because they cannot replicate completely each time the cell divides. For more information about telomeres go to What is a Telomere?
The most significant study about longevity and telomeres was done by Ronald A. DePinho, a Harvard professor, who succeeded in reversing aging in mice. This study was published in Nature. DePinho and his researchers used special mice that had been genetically engineered to age rapidly. The gene that controls aging had been "knocked out”, so these mice had accelerated aging with shrinkage [atrophy] of the brain, spleen, loss of sense of smell, and loss of fertility with testicular atrophy. Callaway: Telomere reverses aging 2010 The next step of the aging mouse experiment was to reverse aging and make the mice younger. This was done by giving back the missing gene that had been “knocked out” and see if that would reverse all these signs of aging in the mice. For this next step, the aged mice were treated with a drug (4-OHT) which dramatically reversed the signs of aging. The aged mice were surprisingly rejuvenated. Their shrunken brains, spleens and testes resumed normal size, and they regained their sense of smell. The infertile males once again became fertile, and fathered large litters. This mice research was a significant breakthrough in controlling the biological clock and postponing aging.
A followup research to the DePinho mice study focused on the major mechanism that controls and activates telomerase ... namely Telomerase reverse transcriptase [ abbreviated to TERT ] or human Telomerase Reverse Transcriptase [ hTERT ] gene. When the hTERT gene is sequenced [ process of determining the order of nucleotide bases (A,C,T, and G) within a stretch of DNA ], and the code examined, one finds two estrogen receptor elements in this gene. This explains why 17-beta estradiol activates telomerase. The fact that there are estrogen receptors in the hTERT gene means that estrogen activates telomerase. Estrogen blockers such as Tamoxifen™ block these receptors and turn off telomerase. Androgens [ e.g. testosterone] were also found to turn on the hTERT gene and activate telomerase, and as expected, androgen blocker drugs inhibited telomerase.
The significance of all these hTERT studies is that one's lifestyle can speed up or slow down the aging process. It stands to reason that many of the strategies that lead to optimal health would also slow down telomere shortening, and research has shown this to be true. Conversely, your lifestyle can also accelerate telomere shortening, effectively causing premature aging. Obesity, lack of exercise, psychological stress and smoking all cause production of free radicals, which can significantly speed up the telomere-shortening process. UCSF: Exercise prevents telomereshortening 2012 But that's not all.
 A
sideline finding of the research about telomerase has opened a new way of thinking
about healing. Studies [ 2008 ] in mice suggest that telomerase also participates in chromosomal repair, since new synthesis of telomere repeats may occur at double-stranded breaks.
This telomerase type of possible therapy can potentially restore organ function and reverse degenerative disease in the the young and elderly.
A
sideline finding of the research about telomerase has opened a new way of thinking
about healing. Studies [ 2008 ] in mice suggest that telomerase also participates in chromosomal repair, since new synthesis of telomere repeats may occur at double-stranded breaks.
This telomerase type of possible therapy can potentially restore organ function and reverse degenerative disease in the the young and elderly.
This may sound confusing but a view of how chromosomes divide can help. Chromosomes get shorter each time the cell divides and as we age, the caps at the end of the chromosomes lose their ability to protect and the cells go into a state called senescence [ cells lose function and grow old ] when they stop dividing. It is believed that this contributes to aging. At conception, our telomeres start out 15,000 base pairs (bps) long and by birth the embryo has divided so many times that the telomere length is down to 10,000 bps. Over the rest of our lifetime we lose another 5,000 bps and when they get down to roughly 3,000 – 5,000bps most of us will be dead.
However, over 90% of the cancers require telomerase to divide continuously. So turning off the telomerase to fight cancer means a trade-off between cancer and aging. Recently, two papers published in Science showed that the mutations in regulatory, non-coding regions of
hTERT gene may be at the root of most melanomas. Together, both independent studies have come up with almost similar frequency of mutations in the promoter region of
hTERT gene
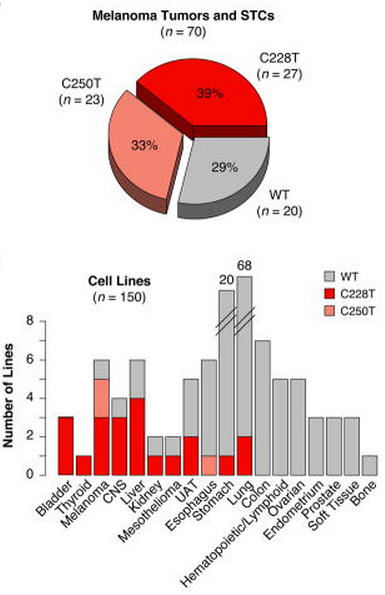
TERT promoter mutations in melanoma are displayed in the pie chart and cancer cell lines in the bar graph below:
It is only a matter of time before researchers identify specific TERT non-coding mutations for other cancers, cardiovascular diseases and other illnesses. Such discoveries become significant when telomerase is blocked in cancer cells as a new therapy with curable implications. The current paradigm holds that chronic infection accelerates erosions of T cell telomeres through increased proliferative stress.
| Pie chart of C228T and C250T somatic mutation status in 70 surveyed melanoma tumors and short-term cultures. Sum of percentages is greater than 100% due to rounding. Bar plot of 150 cancer cell lines of the Cancer Cell Line Encyclopedia (3) depicting TERT promoter mutation status. Individual bars represent the total number of cell lines of a given tumor type interrogated for C228T and C250T mutations, with mutation status indicated by colors defined in the legend. Huang: TERT & melanoma 2013 |
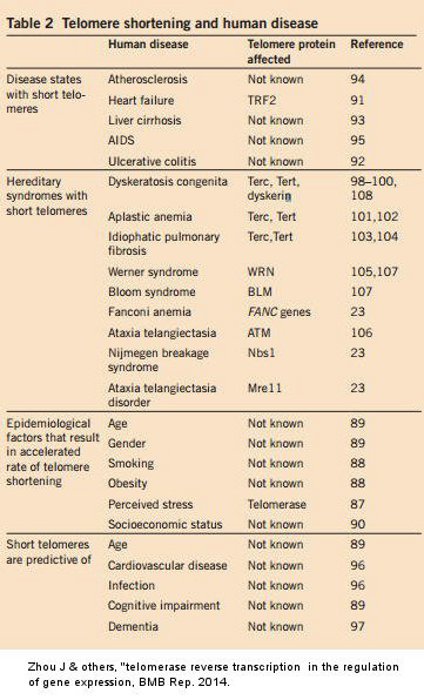
Telomeric length is now recognized as an index of cellular aging and over the last few years measuring of telomeres in peripheral mononuclear cells or in whole blood samples has been applied to patients with a multitude of disease states. Much attention has been given to individuals with atherosclerotic disease and, as a common rule, telomeric length of PBMCs seems to be a strong predictor for disease progression. For example, those with myocardial infarction have shorter telomeres compared to controls Maubaret: Telomeres shorter on heart attack 2010. Leukocyte telomere length is associated with measures of subclinical atherosclerosis. Panayiotou: Telomeres linked to atherosclerosis 2010
Cellular aging reflected by shorter leukocyte telomere length is a predictor for advanced atherosclerosis and cardiovascular disease risk Willeit: Telomere length linked to CVD 2010 . Furthermore, reduced leukocyte telomere length has been linked to increased coronary artery calcium Diaz: Lifesrtyle, telomeres & coronary calcium 2010
and reduction in left ventricular mass Kuznetsova: telomere length & heart size 2010 . However, premature loss of telomeres extends beyond blood vessel disease and emerges as a marker of overall functionality. Numerous studies confirm the link between telomerase and cardiovascular diseases. Diaz: Lifesrtyle, telomeres & coronary calcium 2010 Kuznetsova: telomere length & heart size 2010 Kalpouzos Telomerase link to CVD & cancer 2014 Maubaret: Telomeres shorter on heart attack 2010 Panayiotou: Telomeres linked to atherosclerosis 2010 Willeit: Telomere length linked to CVD 2010
\
Likewise, virus infection seems to be a contributor to immune cell aging and telomere loss. For example, Epstein-Barr virus infection has been described to be associated with telomeric loss Hathcock: Telomerase causes immune system adaptation 1998. Another herpes virus, Cytomegalovirus, may have a particular role in driving immune aging. Hohensinner Telomere Dysfunction, Autoimmunity and Aging 2011
Andrews speculates about the impact of telomere dysfunction in autoimmune and chronic inflammatory disease. Andrews: Telomeres and immunological diseases 2010
In summary, the known potential of telomerase therapy to fight all kinds of diseases is barely the top of the wellness iceberg. Envision cancer, autoimmune disorders and all the other current diseases medical science has not as yet answers for being cured, being treated by the enzyme telomerase. And yes, using such healing technology to solve such problems would probably open a host of other longevity-survival problems. Do not be dismayed, for life is about solving problems and we humans are pretty good at it. This is an exciting dream of the future and it could be here in the next 10 years!
Since time memorial, humans have coveted and sought the fabled "fountain of youth". The good news is that scientists have known for over 50 years the secret of youthfulness and treating diseases successfully. Longevity and good health are linked to telomeres and the enzyme telomerase.
References:
Andrews NP, Fujii H, Goronzy JJ, Weyand CM., "Telomeres and immunological diseases of aging. Gerontology." 2010;56:390–403. Andrews: Telomeres and immunological diseases 2010
Blasco Maria A., "Telomere length, stem cells and aging," NATURE CHEMICAL BIOLOGY, October, 2007, Colume 3 Number 10. Article by Blasco: Telomere length & stem cells is no longer active.
Callaway Ewen, "Telomerase reverses ageing process Dramatic rejuvenation of prematurely aged mice hints at potential therap," Nature, November 28, 2010. Callaway: Telomere reverses aging 2010
Cerni C1, "Telomeres, telomerase, and myc. An update," Mutat Res. 2000 Jan;462(1):31-47. Cerni telomerase cancer update 2000
Cong Yu-Sheng , Woodring E. Wright, and Jerry W. Shay, "Human Telomerase and Its Regulation," Microbiol Mol Biol Rev. 2002 Sep; 66(3): 407–425. Cong: Telomerase regulation 2002
Dach Jeffrey, "Chapter 54: Anti-aging breakthrough with bioidentical hormones," Bioidentical Hormones 101 Book, 2015. Bach: bioidentical hormones & aging 2015
Diaz VA, Mainous AG, 3rd, Everett CJ, Schoepf UJ, Codd V, Samani NJ., "Effect of healthy lifestyle behaviors on the association between leukocyte telomere length and coronary artery calcium," Am J Cardiol. 2010;106:659–63. [PubMed] Diaz: Lifesrtyle, telomeres & coronary calcium 2010
Kuznetsova T, Codd V, Brouilette S, Thijs L, Gonzalez A, Jin Y, Richart T, Van Der Harst P, Diez J, Staessen JA, Samani NJ., "Association between left ventricular mass and telomere length in a population study," Am J Epidemiol, 2010;172:440–50. Kuznetsova: telomere length & heart size 2010
Giuliano Vince, "Telomerase update –arguments for and against using telomere extender supplements," AgeingSciences, May 6, 2012. Giuliano: Pros-Cons of telomerase supplements 2012
Hathcock KS, Weng NP, Merica R, Jenkins MK, Hodes R., "Cutting edge: antigen-dependent regulation of telomerase activity in murine T cells," J Immunol. 1998;160:5702–6. Hathcock: Telomerase causes immune system adaptation 1998
Hohensinner Philipp J., Jörg J. Goronzy, and Cornelia M. Weyand, "Telomere Dysfunction, Autoimmunity and Aging," Aging Dis. December 2. 2011,(6): 524–537. Hohensinner Telomere Dysfunction, Autoimmunity and Aging 2011
Horn S. et al., “TERT promoter mutations in familial and sporadic melanoma,” Science, Feb 22, 2013; 339(6122):959-61. Horn: telomerase & cancer 2013 [ CDKN2A or CDK4, two known melanoma genes ]
Huang F.W. et al., “Highly recurrent TERT promoter mutations in human melanoma,” Science, Feb 22, 2013; 339(6122):957-9. Huang: TERT & melanoma 2013
Kalpouzos Grégoria, Debora Rizzuto1, Lina Keller1, Johan Fastbom1, Giola Santoni, Sara Angleman, Caroline Graff, Lars Bäckman and Laura Fratiglioni, "Telomerase Gene (hTERT) and Survival: Results From Two Swedish Cohorts of Older Adults," The Journal of Gerontology, October 24, 2014 Kalpouzos Telomerase link to CVD & cancer 2014
Maubaret CG, Salpea KD, Jain A, Cooper JA, Hamsten A, Sanders J, Montgomery H, Neil A, Nair D, Humphries SE. "Telomeres are shorter in myocardial infarction patients compared to healthy subjects: correlation with environmental risk factors," J Mol Med. 2010;88:785–94. Maubaret: Telomeres shorter on heart attack 2010
Mitteldorf Josh, "Tomorrow’s Anti-Aging Therapy, Available Today," March 18, 2015. Mitteldorf Anti-aging therapy 2015
Ornish Dean, "Changes in prostate gene expression in men undergoing an intensive nutrition and lifestyle intervention," PNAS, June 17, 2008, vol. 105, no. 24. Ornish: Life style intevention in prostate gene 2008
Ornish Dean, "Intensive Lifestyle Changes for Reversal of Coronary Heart Disease," JAMA, December 16, 1998—Vol 280, No. 23. Ornish: Lifestyle intervention & heart disease 1998
Ornish Dean, "Telomerase and the benefits of healthy living," in Emmanuel Skordalakes Gene Expression and Reg, page 1048, Vol 9 November 2008. Ornish: Telomerase & healthy living 2008
Otis Greg, "Research Targets Melanoma Genetics," Cancer Therapy Advisor, March 27, 2013. Otis: TERT & melanoma genertics 2013
Panayiotou AG, Nicolaides AN, Griffin M, Tyllis T, Georgiou N, Bond D, Martin RM, Hoppensteadt D, Fareed J, Humphries SE,"Leukocyte telomere length is associated with measures of subclinical atherosclerosis," Atherosclerosis. 2010;211:176–81. Panayiotou: Telomeres linked to atherosclerosis 2010
Puterman E1, Lin J, Blackburn E, O'Donovan A, Adler N, Epel E., "The power of exercise: buffering the effect of chronic stress on telomere length," PLoS One. 2010 May 26;5(5). Puterman: Exercise buffers telomere length 2010
Sears, Al, "Transcript of "Telomere seminar dec2012_new," Dr. Sears Center for Health & Wellness, Aug 19, 2014. Article by Sears: Telomere update is no longer acailable.
Telomerase reverse transcriptase (abbreviated to TERT, or hTERT in humans) is a catalytic subunit of the enzyme telomerase, which, together with the telomerase RNA component (TERC), comprises the most important unit of the telomerase complex.[1][2]
Telomerases are part of a distinct subgroup of RNA-dependent polymerases. Telomerase lengthens telomeres in DNA strands, thereby allowing senescent cells that would otherwise become postmitotic and undergo apoptosis to exceed the Hayflick limit and become potentially immortal, as is often the case with cancerous cells.
Background: Some simplified facts:
- Telomeres and telomerase are relatively new, important and dynamic areas of aging science.
- Telomeres are caps at the end of chromosomes.
- Telomerase is a naturally-occurring enzyme which lengthens telomeres when activated. Germ cells and stem cells express relatively high levels of telomerase. Many normal body cells express little or no telomerase.
- Each time a cell divides the telomeres get a little shorter due to the mechanics of cell division.
- With aging after a certain number of cell divisions, telomeres in a given cell get critically short. Older people generally have shorter telomeres. Diseases, stress and a number of other conditions can also cause telomeres to shorten.
- Cells with too-short telomeres can become senescent or suffer apoptosis (die).
Tosato Matteo and others, "The aging process and potential interventions to extend life expectancy," Clin Interv Aging. September 02, 2007 2(3): 401–412. Tosato: Aging theories 2007
UCSF, "Exercise may prevent stress on telomeres, a measure of cell health," EurikaAlert, University of California San Francisco, April 4, 2012. UCSF: Exercise prevents telomere shortening 2012
Wade Nickolas, "3 Americans Share Nobel for Medicine," The New York Times, October 5, 2009. Article about Wade: Nobel prize Telomerase is no longer active.
Willeit P, Willeit J, Brandstatter A, Ehrlenbach S, Mayr A, Gasperi A, Weger S, Oberhollenzer F, Reindl M, Kronenberg F, Kiechl S., "Cellular aging reflected by leukocyte telomere length predicts advanced atherosclerosis and cardiovascular disease risk," Arterioscler Thromb Vasc Biol. 2010;30:1649–56. Willeit: Telomere length linked to CVD 2010
Yang Lili, "Naïve T-cells in Myelodysplastic Syndrome Display Intrinsic Human Telomerase Reverse Transcriptase (hTERT) Deficiency," Leukemia. 2013 Apr; 27(4): 897–906. Yang: hTERT deficiency in MDS 2015
Zhou Junzhi , Deqiang Ding, Miao Wang & Yu-Sheng Cong, "Telomerase reverse transcriptase in the regulation of gene expression," BMB Rep. 2014; 47(1): 8-14. Article by Zhou hTRET regulates gene expression is no longer active.